Supercritical Fluids Extraction: Benefits and Applications
Overview
Supercritical fluids extraction (SFE) utilizes supercritical fluids to extract valuable compounds from various materials efficiently. This article explains how SFE works, its key advantages, and its applications in food, pharma, and cosmetics.
Key Takeaways
Supercritical fluids extraction (SFE) utilizes CO2 and other fluids at high temperatures and pressures to extract valuable compounds from various raw materials efficiently.
SFE offers significant advantages over traditional methods, including faster extraction times, reduced solvent usage, and a lower environmental impact due to minimal residue and non-toxic solvents.
Key parameters such as temperature, pressure, and the use of co-solvents play critical roles in optimizing the SFE process for enhanced efficiency and yield across diverse industries.
Understanding Supercritical Fluids Extraction
The extraction technique utilizing supercritical fluids, known as supercritical fluid extraction (SFE), leverages the distinct characteristics of these fluids to isolate valuable substances from raw materials. When a substance is exposed to conditions surpassing its critical point in terms of temperature and pressure, it transitions into a supercritical phase where the properties of liquid and gas phases merge.
As a result, this generates a solvent that possesses a permeating capability similar to that of a gas, along with the dissolving power of a liquid. Such attributes render it particularly effective for extracting purposes, which is why this method is referred to as “supercritical extraction.’’
Below is a diagram of the supercritical point of CO2.
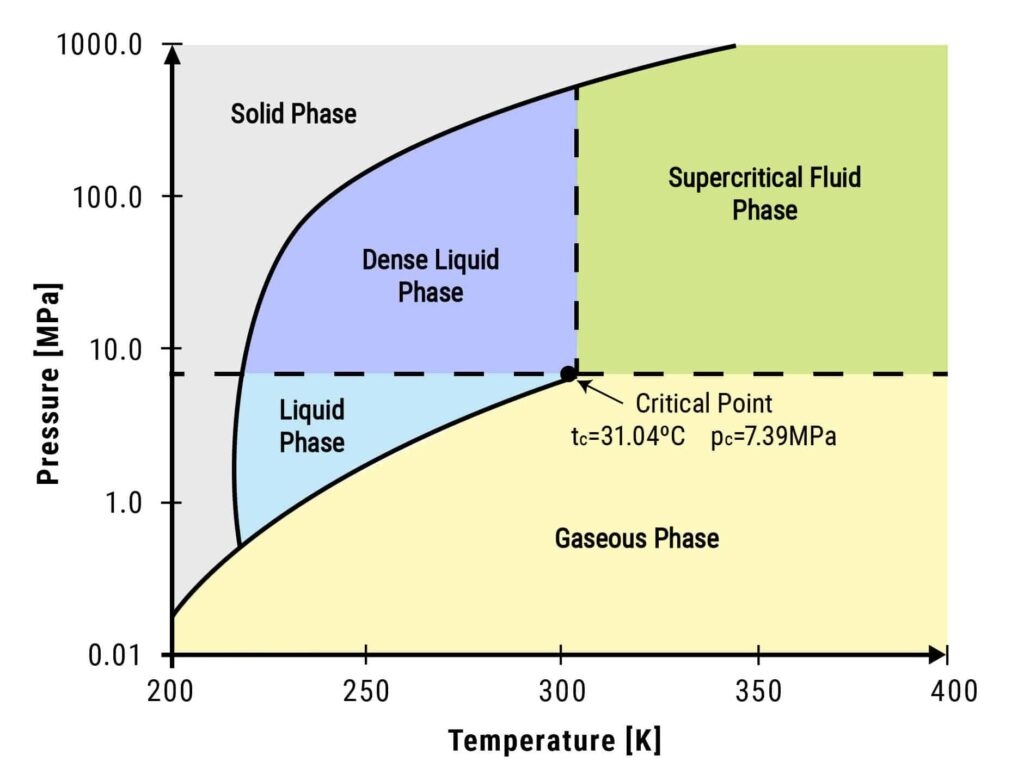
Definition and Principles
At the core of supercritical fluid extraction (SFE) is the concept of supercritical fluid, where a substance demonstrates characteristics similar to both gases and liquids. Attaining this supercritical state involves subjecting a fluid to conditions beyond its critical point with increased heat and pressure.
This creates a phase capable of effortlessly permeating through solid structures while proficiently dissolving substances, thereby addressing limitations associated with mass transfer and making these fluids exceptionally suitable for compound extraction from various sources.
Common Supercritical Fluids
Carbon dioxide (CO2) is the most popular supercritical fluid utilized in extraction processes due to its relatively low critical temperature and pressure, simplifying attaining and retaining a supercritical state. CO2’s non-toxicity, non-flammability, and accessibility from existing industrial activities also enhance its desirability while minimizing its environmental footprint.
Supercritical fluid extraction with CO2 involves isolating compounds ranging from essential oils to bioactive substances. Its capacity to serve as an effective solvent without leaving harmful residues makes it an optimal option within food production, pharmaceuticals, and cosmetics industries.
Alternate supercritical fluids are used tailored to particular needs for other types of extractions. This includes water amongst various organic solvents. Supercritical water has been used to break down hazardous waste via methods like supercritical water oxidation or converting biomass into valuable fuels, demonstrating another aspect where these powerful solvents are implemented.
Advantages of Supercritical CO2 Extraction

Supercritical CO2 extraction presents numerous benefits when compared to traditional extraction techniques. Its efficiency stands out as a key advantage, being capable of performing extractions up to 25 times faster than the conventional Soxhlet method, thereby greatly diminishing the duration required for processing.
This approach is more efficient in using resources and cost-effective since it requires up to 30 times less solvent than standard methods.
Efficiency and Selectivity
Supercritical CO2 extraction stands out for its superior efficiency and selectivity. Due to this method’s elevated diffusivity and mass transfer properties, it provides quicker and more effective results than conventional liquid extraction techniques.
Adding detergents that dissolve in carbon dioxide can improve the solvating capability of supercritical CO2. This adjustment leads to a more targeted selection during the extraction process, isolating desired compounds with greater precision.
Environmental Benefits
Supercritical carbon dioxide extraction is acclaimed for its effectiveness and eco-conscious nature. The fact that supercritical CO2 is neither flammable nor toxic positions it as a superior choice over traditional organic solvents in terms of safety. This technique significantly reduces solvent residue within the end product, lessening ecological harm.
Key Parameters in Supercritical Fluids Extraction
The effectiveness of extraction using supercritical fluids is significantly determined by crucial factors, including temperature, pressure, and the incorporation of co-solvents at critical points. These factors have a direct impact on both the solubility and the amount extracted for various compounds.
It’s essential to understand and fine-tune these parameters to enhance the productivity of the extraction process within industrial settings.
Temperature and Pressure Effects
In supercritical fluid extraction, both temperature and pressure are crucial factors. Elevating the pressure typically boosts compound solubility due to increased fluid density. Critical pressure significantly influences how effectively this process is carried out.
In a complementary manner, raising the temperature can lead to higher vapor pressures of solutes, which might amplify yields from extractions even though it may simultaneously decrease solvent density.
Introducing co-solvents into the mix can potentially diminish both the critical temperature and pressure necessary for successful extraction operations.
Role of Co-Solvents
Co-solvents are vital in modifying the polarity of the extraction medium, facilitating the extraction of more polar substances. Ethanol and methanol are commonly used co-solvents that significantly enhance the solubility of polar compounds in supercritical fluid extraction.
Co-solvents significantly improve the efficiency of the extraction process, playing a crucial role in achieving high yields and purity of the extraction solvent materials.
Applications of Supercritical Fluids Extraction

Many industries, from food and pharmaceuticals to cosmetics, employ supercritical fluid extraction for its high efficiency and flexible applications. This method stands out as an optimal technique for isolating precious substances.
The utilization of supercritical fluid extraction in pivotal sectors underscores its capacity to adapt and perform effectively across various contexts.
Food Industry
Within the food industry, isolating valuable compounds from plants is commonly achieved using supercritical CO2 extraction. This technique excels in retrieving essential oils and antioxidants, improving both the quantity and quality of these extracted substances.
In the realm of food science, employing supercritical CO2 extraction is especially advantageous as it guarantees that the materials are devoid of any toxic solvent remnants. This method is favored when producing superior-grade food components.
Pharmaceutical Industry
Supercritical fluid extraction (SFE) offers substantial advantages to the pharmaceutical industry by efficiently extracting active ingredients from natural sources, thereby improving their solubility and bioavailability. This technique guarantees that the compounds preserve their bioactive characteristics, enhancing therapeutic outcomes and heightened efficacy in pharmaceutical use.
Cosmetics Industry
The cosmetics sector prefers to use supercritical fluid extraction to acquire natural ingredients, enhancing the quality and appeal of their products. The technique protects delicate compounds throughout the extraction process, delivering pure natural components without introducing any harmful solvents.
Employing supercritical CO2 in this method facilitates the recovery of superior essential oils and various organic compounds. This advancement elevates the standard excellence observed within cosmetic goods.
Case Studies in Supercritical Fluids Extraction

Let’s examine some case studies to grasp the real-world uses and advantages of Supercritical Fluid Extraction (SFE). By looking at these instances, we can see how SFE improves product quality and efficiency in various sectors, highlighting its adaptability and efficacy.
Extraction of Piperine from Peppers
A case study recently showcased the practical separation of piperine from peppers through supercritical CO2 extraction. The distinctive characteristics of supercritical CO2, including its adjustable solubility, were key in obtaining a high yield and pure extract.
This efficient extraction technique is crucial for creating premium piperine that can be used across numerous sectors like food production, pharmaceuticals, and cosmetic manufacturing.
Extraction of Bisphenol A from Epoxy Resin
Another case study demonstrated the extraction of Bisphenol A from epoxy resin using supercritical fluid extraction with CO2 at a pressure of 25 MPa. This method allowed for the efficient recovery and analysis of Bisphenol A, highlighting the effectiveness of SFE in removing unwanted compounds from complex matrices.
Innovations and Future Trends in Supercritical Fluids Extraction
The field of supercritical fluid extraction is continuously evolving, with emerging trends and innovations pushing the boundaries of what is possible. From environmental applications to material science advancements, the future of SFE is promising.
This section will explore some of these exciting developments.
Nano and Micro-Particle Formation
The utilization of supercritical fluids is becoming more prevalent for obtaining controlled particle sizes at the nanometer scale, improving the materials’ characteristics. Adjusting variables such as pressure and temperature enables the customization of particle traits, providing superior control over particle size compared to traditional techniques.
Such proficiency in manipulating particle dimensions is especially advantageous within the realm of material science, where exacting control over particles is essential.
Supercritical Water Oxidation
Another inventive use of Supercritical Fluid Extraction (SFE) is found in supercritical water oxidation, which primarily serves to process toxic waste. By functioning at increased temperatures and pressures, this method accelerates reaction rates while guaranteeing the protection of the environment.
Supercritical water gasification can break down biomass into hydrocarbon fuels or hydrogen, significantly advancing energy sustainability.
Practical Considerations for Implementing Supercritical Fluids Extraction
Equipment Selection and Setup
It’s critical to consider the operational scale and unique properties of the substances being processed when selecting equipment for supercritical fluid extraction. The M-2.5 ScCO2 Extractor by extraktLAB is well-suited for small-scale producers or research laboratories and acts as an efficient vessel for extraction.
For larger production demands, the E-180 ScCO2 Extractor stands out as it is recognized as the quickest supercritical CO2 extractor available, enhancing productivity levels. extrakrLAB offers extensive support that covers commissioning, training, and maintenance assistance to guarantee optimal functionality and operation safety.
Summary
To summarize, utilizing supercritical fluids extraction stands out as an advantageous substitute for traditional extraction techniques, delivering enhanced efficiency, specificity, and ecological advantages. With technological advancements, the prospect of SFE is optimistic, as breakthroughs lay the groundwork for novel uses and refining existing procedures.
Frequently Asked Questions
What are the disadvantages of supercritical fluid extraction?
The disadvantages of supercritical fluid extraction (SFE) primarily include high costs associated with equipment and operation and limitations in extracting polar compounds due to the non-polar nature of CO2.
What is the primary advantage of supercritical CO2 extraction over conventional methods?
The primary advantage of supercritical CO2 extraction is its significantly higher efficiency and faster extraction times while utilizing up to 30 times less solvent than conventional methods.
Why is supercritical CO2 considered environmentally friendly?
Supercritical CO2 is environmentally friendly because it is non-flammable, non-toxic, and frequently derived from industrial processes, minimizing its environmental footprint and leaving little to no solvent residue.
Contact Us To Get Started Today







